Classification of Proteins
Level of Organisation
i) Primary Structure
-the sequence of amino acids in the polypeptide chain
-linked by peptide bonds

ii) Secondary Structure
-the folding of polypeptide chains into specific shape
- α -helix: coiled polypeptide chain
- β -pleated: folded polypeptide chain
-hydrogen bonds hold the peptide chains in place
-eg:keratin:present in hair and nails

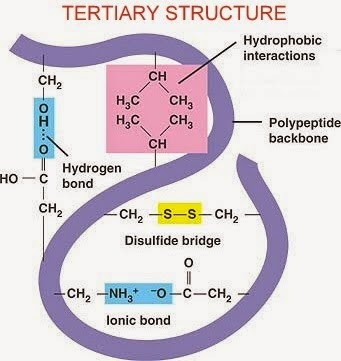
iii. Tertiary structure
-protein is folded into a three-dimensional shape
-shape is stabilized by hydrogen bonds,hydrophobic intereactions,disulphide bridges and ionic bonds
-eg:enzyme,myoglobin
iv. Quartenary Structure
-more than one polypeptide chain
-forms a three dimensional shape
-hydrogen bonding,ionic bonds,hydrophobic interactions holds maintains its shape
-eg:haemoglobin,an oxygen carrying molecule which is made up of 2 α -helix and 2 β -pleated polypeptides,consists of 4 iron haem group for oxygen binding.
Structure of haemoglobin

Fibrous vs Globular Protein
