
What Are Atomic Number and Atomic Weight?
What is an element?
Chemical behavior is what originally led scientists to classify matter into various elements- Interactive periodic table with up-to-date element property data collected from authoritative sources. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!
- An isotope is one of two or more species of atoms of the same chemical element that have different atomic mass numbers (protons + neutrons). The atomic weight of helium is 4.002602, the average that reflects the typical ratio of natural abundances of its isotopes. Atomic weight is measured in atomic mass units (amu), also called daltons.
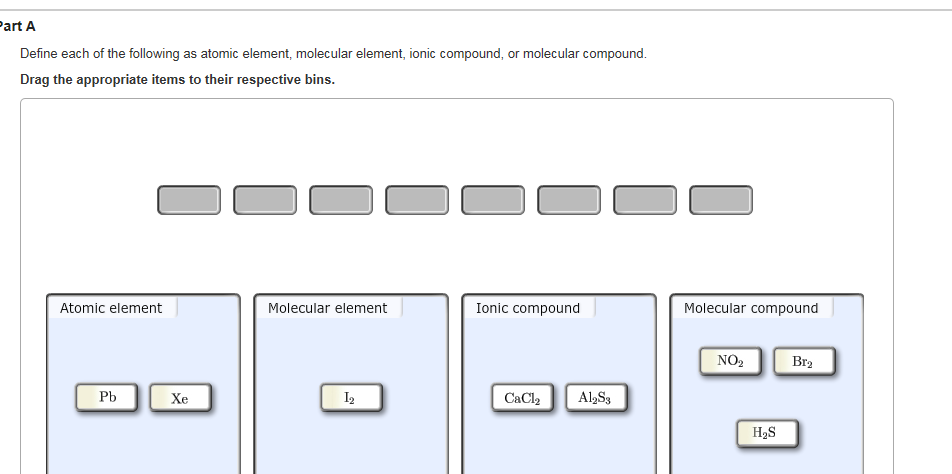
An atomic-level view of elements and compounds. Discussed in this video are atomic elements, molecular elements, molecular compounds, and ionic compounds.
. Chemical behavior is the ability of an atom to combine with other atoms. In more technical terms, chemical behavior depends upon the type and number of the chemical bonds an atom can form with other atoms. In classroom kits for building models of molecules, atoms are usually represented by colored spheres with small holes for pegs and the bonds are represented by the small pegs that can connect the spheres. The number of peg holes signifies the maximum number of bonds an atom can form; different types of bonds may be represented by different types of pegs. Atoms that have the same number of peg holes may have similar chemical behavior. Thus, atoms that have identical chemical behavior are regarded as atoms of the same element. For example, an atom is labeled a 'carbon atom' if it can form the same number, types, and configurations of bonds as other carbon atoms. Although the basics are simple to explain, how atoms bind to each other becomes very complex when studied in detail; new discoveries are still being made as new types of materials are formed.What is atomic number?
An atom may be visualized as a miniature solar system, with a large central nucleus orbited by small electrons. The bonding capacity of an atom is determined by the electrons. For example, atoms that in their normal state have one electron are hydrogen atoms and will readily (and sometimes violently) bond with oxygen. This bonding capacity of hydrogen was the cause of the explosion of the airship Hindenburg in 1937. Atoms that in their normal state have two electrons are helium atoms, which will not bond with oxygen and would have been a better choice for filling the Hindenburg.We can pursue the question back one step further: What determines the number of electrons? The number of protons in the nucleus of the atom. Here, the analogy between an atom and the solar system breaks down. The force that holds the planets in their orbits is the gravitational attraction between the planets and the sun. However, in an atom what holds the electrons in their orbit is the electrical attraction between the electrons and the protons in the nucleus. The basic rule is that like charges repel and opposite charges attract. Although a proton has more mass than an electron, they both have the same amount of electrical charge, but opposite in kind. Scientists have designated electrons as having a negative charge and protons as having a positive charge. One positive proton can hold one negative electron in orbit. Thus, an atom with one proton in its nucleus normally will have one electron in orbit (and be labeled a hydrogen atom); an atom with ninety-four protons in its nucleus will normally have ninety-four electrons orbiting it (and be labeled a plutonium atom).
The number of protons in a nucleus is called the atomic number and always equals the number of electrons in orbit about that nucleus (in a nonionized atom). Thus, all atoms that have the same number of protons--the atomic number--are atoms of the same element.
What is atomic weight?
The nuclei of atoms also contain neutrons, which help hold the nucleus together. A neutron has no electrical charge and is slightly more massive than a proton. Because a neutron can decay into a proton plus an electron (the essence of beta decay), it is sometimes helpful to think of a neutron as an electron and a proton blended together, although this is at best an oversimplification. Because a neutron has no charge, a neutron has no effect on the number of electrons orbiting the nucleus. However, because it is even more massive than a proton, a neutron can add significantly to the weight of an atom. The total weight of an atom is called the atomic weight. It is approximately equal to the number of protons and neutrons, with a little extra added by the electrons. The stability of the nucleus, and hence the atom's radioactivity, is heavily dependent upon the number of neutrons it contains.What notations are used to represent atomic number and weight?
 Each atom, therefore, can be assigned both an atomic number (the number of protons equals the number of electrons) and an atomic weight (approximately equaling the number of protons plus the number of neutrons). A normal helium atom, for example, has two protons and two neutrons in its nucleus, with two electrons in orbit. Its chemical behavior is determined by the atomic number 2 (the number of protons), which equals the normal number of electrons; the stability of its nucleus (that is, its radioactivity) varies with its atomic weight (approximately equal to the number of protons and neutrons). The most well-known form of plutonium, for example, has an atomic number of 94, since it has 94 protons, and with the 145 neutrons in its nucleus, an atomic weight of 239 (94 protons plus 145 neutrons). In World War II, its very existence was highly classified. A code number was developed: the last digit of the atomic number (94) and the last digit of the atomic weight (239). Thus, in some of the early documents examined by the Advisory Committee, the term 49
Each atom, therefore, can be assigned both an atomic number (the number of protons equals the number of electrons) and an atomic weight (approximately equaling the number of protons plus the number of neutrons). A normal helium atom, for example, has two protons and two neutrons in its nucleus, with two electrons in orbit. Its chemical behavior is determined by the atomic number 2 (the number of protons), which equals the normal number of electrons; the stability of its nucleus (that is, its radioactivity) varies with its atomic weight (approximately equal to the number of protons and neutrons). The most well-known form of plutonium, for example, has an atomic number of 94, since it has 94 protons, and with the 145 neutrons in its nucleus, an atomic weight of 239 (94 protons plus 145 neutrons). In World War II, its very existence was highly classified. A code number was developed: the last digit of the atomic number (94) and the last digit of the atomic weight (239). Thus, in some of the early documents examined by the Advisory Committee, the term 49Atomic Element Definition
refers to plutonium.Styles of notation vary, but usually isotopes are written as:
atomic number Chemical abbreviation atomic weight
or as
atomic weight Chemical abbreviation
Atomic Element Examples
Thus, the isotope of plutonium just discussed would be written as:
94Pu239
or as
239Pu
Atomic Number On Periodic Table
Since the atomic weight is what is often the only item of interest, it might also be written simply as Pu-239, plutonium 239, or Pu239.
